By Marissa SteplerCommonly known as the “powerhouses” of the cell, mitochondria provide the vast majority of energy required for cells to function, grow, move, and thrive. Mitochondria self-replicate and have their own DNA, but only 37 of the approximately 3000 genes needed for mitochondrial function are found in mitochondrial DNA. The majority of genes contributing to mitochondrial energy generation, nucleotide synthesis, hormone production, and the many other functions of the mitochondria are located in the nuclear chromosomal DNA of each cell.
When the “powerhouses” of the cell fail, cellular health and function can be drastically decreased, often leading to cell dysfunction and death across numerous different cell types and tissues. In mitochondrial disease, this dysfunction occurs due to inherited or spontaneous mutations in nuclear genes encoding proteins involved in mitochondrial function or mitochondrial DNA. Mutations in nuclear genes can be inherited from either parent. However, because, in a fertilized embryo, all cytoplasm and mitochondria are provided by the egg, mutations in the mitochondrial DNA of a child are inherited solely from his mother. Furthermore, a mother’s mitochondrial DNA mutation may occur in a small enough percentage of her total mitochondria that she remains unaffected, yet the mutation may be lethal when passed on to her children. So if a mother knows she carries a mitochondrial DNA mutation, how can she prevent the transmission of the mutation to her children? Recently, researchers have addressed this problem through a new variation on in vitro fertilization (IVF) called three-parent IVF. In this procedure, instead of using an egg from a single female donor and sperm from a single male donor, eggs from two female donors – one from the mother and one from a female donor lacking mitochondrial DNA mutations – are used. When a cell, such as an egg cell, undergoes DNA replication, the dividing chromosomes are pulled apart by microtubule spindles before being separated into two daughter nuclei. In three-parent IVF, a technique called spindle transfer is used to extract the spindles and their attached chromosomes, which are then injected into the healthy donor egg from which the spindles have been removed. This new combined egg is then fertilized with the father’s sperm, resulting in an embryo which has maternal and parental nuclear DNA but healthy donor mitochondrial DNA. While this IVF method has been previously tested in primates and mammals, it was not attempted in humans until recently, when the procedure was used by a mother who had a mitochondrial gene mutation which results in Leigh syndrome, a lethal neurological disorder. After two of her previous children inherited the disease, the mother underwent spindle transfer three-parent IVF, which resulted in a successful pregnancy and birth of a healthy baby boy. Although considered controversial in some circles, three-parent IVF seems to be a powerful and effective method for preventing some inherited human mitochondrial diseases. As technology continues to advance, this procedure may be used to prevent other inherited disorders related to mitochondria and organelle dysfunction. Currently, this breakthrough procedure offers hope for parents carrying mutations in mitochondrial disease-related genes and for scientists researching organelle-related diseases. Sources: [1] https://www.sciencenews.org/article/three-parent-babies-explained [2]http://www.umdf.org/site/c.8qKOJ0MvF7LUG/b.7934627/k.3711/What_is_Mitochondrial_Disease.htm [3]http://whut.pbslearningmedia.org/resource/tdc02.sci.life.cell.mitochondria/the-powerhouse-of-the-cell/
0 Comments
by Angela Wong Credit: Latin American Culture Club Credit: Latin American Culture Club Recent discoveries may help shed light on what makes us all unique, and why even identical twins can be so unlike (Salk, 2016). These findings have large implications on not only what distinguishes us from one another, but also on what sets Homo sapiens apart from other eukaryotes. In a sense, these factors simultaneously define and unite us. LINE-1 retrotransposons (L1s), also called “jumping genes,” are small pieces of DNA that have been known to insert genetic information throughout the genome. “Jumping” around the genome, they amplify themselves which lead regulatory DNA to be copied and shuffled (Ivancevic, Kortschak, Bertozzi et al., 2016). Present in most healthy neurons, L1s are a source of genomic diversity on the brain. Variations between neurons within the same brain suggest that they “function slightly differently from each other,” says the study’s senior investigator, Rusty Gage. Due to L1s and other factors, each neuron contains around 1,500 unique mutations (Yong, 2015). Diversity of neurons, which process and transmit information in the brain, impacts one’s brain functionality (Salk, 2016). Additionally, at least a third of the 20,000 different genes in the human genome are expressed in the brain—the highest proportion of genes expressed in the human body. According to latest studies, L1s can also remove large chunks of DNA, affecting the genome even more significantly than previously thought. Evidence has shown that neurons from those with schizophrenia and Rett syndrome have above-average levels of L1 variations within their genomes. Researchers at the Salk Institute believe that these findings indicate will further the understanding of the role, and they plan to continue exploring the role of L1 variations and how they impact both brain function and illness (Salk, 2016). Other scientists conducted a “comprehensive phylogenetic analysis of elements from over 500 species from widely divergent branches in the tree of life” to examine the diversity of L1s, which are thought of as “tightly constrained, homologous, and ubiquitous elements with well-characterized domain organization.” They found that in recent times, the growth of L1 elements in mammalian species have diverged from lineages in other plants and metazoans, animals with cell differentiation. Illustrating that “both long-term inherited evolutionary patterns and random bursts of activity in individual species can significantly alter genomes” (Ivancevic et al., 2016). These two studies highlight the significance of L1s in the expression of characteristics that make each one of us unique, as well as uniquely human. Sources: Ivancevic, A. M., Kortschak, D., Bertozzi, T., & Adelson, D. (2016, September 26). LINEs between species: Evolutionary dynamics of LINE-1 retrotransposons across the eukaryotic tree of life. Genome Biology and Evolution. Retrieved from http://gbe.oxfordjournals.org/content/early/2016/09/30/gbe.evw243.abstract The brain’s stunning genomic diversity revealed - Salk ... (2016, September 9). Retrieved October 21, 2016, from http://www.salk.edu/news-release/brains-stunning-genomic-diversity-revealed/ Yong, E. (2016, October 1). The Surprising Genealogy of Your Brain - The Atlantic. Retrieved October 21, 2016, from http://www.theatlantic.com/science/archive/2015/10/the-genealogy-of-your- brain/408232/ By Marissa Stepler Stem cells have long been a source of controversy. Most of the concern related to the use of stem cells in medical research is rooted in the fact that stem cells have to be sourced from embryonic tissue, such as the umbilical cord. Because many religious groups oppose the use of embryos in stem-cell research, the topic has been hotly debated in recent years. However, recent research has produced what may be a viable medical alternative to embryonic stem cells. The stem cells being used - and showing a lot of promise - in many current research studies are called induced pluripotent stem cells (iPSCs). iPSCs are made by “reprogramming” adult cells by treating them with a mixture of growth factors and other substances, provoking them to acquire some of the characteristics of embryonic cells. The most important feature of iPSCs for their use in medical research is their ability to differentiate into a number of different cell types, including skin cells, muscle cells, and neurons. Because iPSCs can develop into skin, muscle, and nerve cells, the use of iPSCs to treat autoimmune diseases that affect the musculoskeletal and nervous systems, such as rheumatoid arthritis and lupus, is currently being investigated. iPSCs injected into the joints of rheumatoid arthritis patients have the potential to regenerate cartilage damaged or destroyed by the disease, and experimental treatment with iPSCs has allowed some patients crippled by this disease and others affected by severe systemic lupus to resume almost normal functioning. The greatest advantage of iPSC treatment, especially for patients with autoimmune diseases, is that a patient can be treated with cells sourced from his or her own body, reducing the likelihood of adverse reactions to iPSC treatment. As research on adult-derived stem cells continues to advance, the treatment options for autoimmune conditions - even those that we currently consider incurable - are expanding, offering new hope to individuals suffering from diseases like rheumatoid arthritis and lupus.
Sources: [1] http://stemcells.nih.gov/info/basics/pages/basics10.aspx [2] http://www.healthline.com/health-news/stem-cell-therapy-a-possible-treatment-for-rheumatoid-arthritis-010516#4 [3] http://www.americancatholic.org/News/StemCell/ By Marissa SteplerMost of us listen to music frequently in our lives: on the radio, at concerts, and even in coffee shops. Although music has a much richer sound than the spoken word, it has always been thought to be perceived the same way that we perceive other noises. But what if a part of the brain was dedicated solely to the perception and processing of musical stimuli - a “musical cortex”? According to researchers at MIT, the “musical cortex,” or something like it, may actually exist. Dr. Josh McDermott and other scientists at the McGovern Institute for Brain Research at MIT believe that they have found a discrete population of neurons in the auditory cortex that respond solely to musical sounds. McDermott’s team used functional MRI (fMRI) imaging in a new way, using the image data to identify particular groups of neurons that responded to different stimuli. The researchers found six different populations of neurons that respond to unique types of sounds, one of which was music. One of the other populations they identified was responsive to speech and did not overlap with the music population, suggesting that music and speech are processed in different places and probably in different ways by the brain. But if all musical sounds are processed by the same set of neurons, how do we distinguish between Taylor Swift and Beethoven? There is still a lot that is unknown about how these music-specific neurons work, but McDermott and his team think that different subgroups of “music neurons” may respond to musical elements like rhythm and melody. It is also possible that these neurons may function differently in each individual, depending on their childhood exposure to music and musical training (or lack thereof).
So maybe part of the reason we as humans like music so much is because our brains are specially equipped to listen to it. As we learn more about the way the brain processes music, we may gain insight not only into the higher functioning of the auditory cortex, but also about the way that our brains have adapted to the prevalence of music as a dominant part of our culture. Sources: [1] http://news.mit.edu/2015/neural-population-music-brain-1216 [2] http://www.cell.com/neuron/fulltext/S0896-6273(15)01071-5 By Amy MengHold up a piece of paper. What do you see? If you are a Buddhist Zen master, you will clearly see that there is a cloud floating in the sheet of paper. Without the cloud, there is no rain; without rain, there are no trees; without trees, there is no paper. If you look even closer, you will see the paper mill and the logger, and the logger’s daily bread. You will see the sunshine that nourishes wheat for the bread, and the sun that holds the world in perfect orbit. The cloud and the paper “inter-are.” [1]. This lesson in “interbeing” originates from Buddhist monk and peace activist Thích Nhất Hạnh. Whether we know it or not, Hanh's teachings permeate our world. In fact, his autobiographical documentary, “Walk with Me,” will be narrated by none other than Benedict Cumberbatch, who identifies as Buddhist –“at least philosophically" - and attributes much of his success to the teachings of Hạnh. Other prominent figures who claim to have been influenced by Hanh include comedian Garry Shandling and Alejandro Iñárritu, director of the Oscar-winning Birdman and The Revenant [2]. Hạnh teaches us that separateness is an illusion. To be is to inter-be. The impetus of this lesson is especially apparent in light of Earth’s warming climate. We have all heard that permafrost in the arctic is melting very quickly. More unfamiliar is that this permafrost harbors enormous stores of methane gas that, when released, will raise global temperatures at a catastrophic rate. Even more frightening is that some United States presidential candidates don’t accept this as fact.
And so, with election season in full-swing and Earth Day just around the corner, let us reflect on our responsibility as voters. In evaluating our candidates, let us dissect both their social and environmental policies. Yes, charisma and determination are necessary traits of great leaders, but what about open-mindedness and analytical thinking? Are they, so to speak, scientists? Let us hold our candidates up to scientific criticism and consider their attitudes toward our environment. In realizing our interbeing with nature, we realize that we are ever more alike, ever united. The universe does not care what God we worship, or what political party we support. The universe does not care that our beautiful, blue-marbled home is dying. We are the sole protectors of the only planet known to sustain life, and it is our duty to stand united for our common home. Let us vote not as citizens of America, but as citizens of Earth. For, in the wisdom of Thích Nhất Hạnh, to be is to inter-be. Sources [1] http://www.lionsroar.com/the-fullness-of-emptiness/ [2] http://www.lionsroar.com/benedict-cumberbatch-narrate-thich-nhat-hanh-documentary/ [3] https://nsidc.org/cryosphere/frozenground/methane.html By Amy Meng Unless you’ve been living under a rock, you probably know by now that there is water on Mars. You probably also breathed a sigh of disappointment when this water was announced as inhospitably salty, presuming that this news was then crammed into a box somewhere labeled: “NASA’s Anticlimactic Letdowns”. But in space agencies around the world, precisely the opposite happened: champagne bottles popped open as researchers celebrated a monumental triumph for mankind. Though we have yet to find definitive evidence of extraterrestrial life, we now know for certain that Earth is not as rare as we had previously thought in its ability to host life. This opens, quite literally, a whole new universe of riveting possibilities. NASA’s gaze has now shifted to Europa, one of Jupiter’s moons. Initially, Europa seems pretty unfriendly. Its surface is bombarded with dangerous ionizing radiation and encased in a kilometers-thick crust of ice (okay, maybe unfriendly is an understatement). However, researchers using the Hubble Telescope in 2013 have spotted geysers of water blasting out from within its surface. They believe that, aside from tidal action, this water is heated by hydrothermal vents which are also responsible for Earth’s early lifeforms. A source of heat energy, water, and shelter from dangerous radiation—no wonder Europa is NASA’s newest darling. (Jupiter’s moon Enceladus is also quite ravishing, but that’s another story.) In light of these exciting findings, NASA’s Jet Propulsion Laboratory (JPL) is assembling a flyby mission to sample Europa’s geysers in the mid-2020s. The spacecraft will orbit Jupiter and conduct 45 flybys of Europa, also investigating the composition of its interior and icy shell. Regardless of what we find, this mission this will bring us ever closer to answering the question: are we alone in the universe?
One enthusiastic proponent of this mission is William Nye, CEO of the Planetary Society (you probably know him as “Bill Nye the Science Guy,” or “the bowtie dude with wicked eyebrows"). The Planetary Society is the world’s largest nonprofit organization supporting space exploration. In an interview by Big Think, Nye addresses qualms about the expense of the mission. He estimates costs to total approximately $2 billion, a price he jokes is “relatively inexpensive.” “But,” he continues, gesturing excitedly in Science Guy-esque fashion, “$2 billion spread over ten years is barely the cup of coffee per taxpayer – once. And my feeling is that people buy more than one cup of coffee every ten years.” Source: [1] http://www.huffingtonpost.com/2014/08/04/bill-nye-europa-jupiter-extraterrestrial-life-video_n_5638320.html [2] http://www.jpl.nasa.gov/missions/europa-mission/ Click to Donate to The Planetary Society! By Sean Rowell Writer 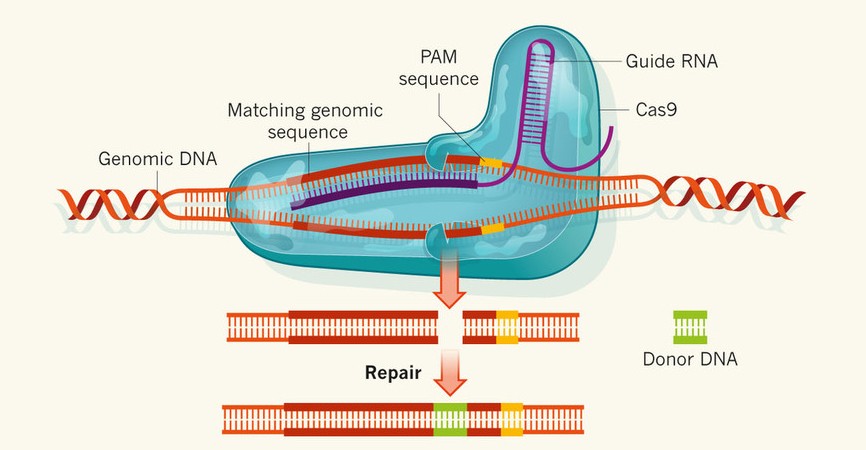 From: Charpentier and Doudna 2013 From: Charpentier and Doudna 2013 Imagine that you had the opportunity to rid the world of disease by preventing the suffering of millions through one single treatment. Would you do it? This question, once a far-fetched idea, is becoming a reality as gene therapy techniques become more advanced and precise. Gene therapy is a branch of science that involves manipulating genes to treat a disease or even prevent one from occurring in the first place. There are many ways in which this can be done. A researcher may replace a gene that is not functioning properly and causing disease with a healthy version of the gene. This researcher could also introduce a brand new gene into the patient that would help fight a certain disease. Whatever the method, this type of gene therapy offers a way for patients to be treated for their diseases at the source. Instead of merely treating the symptoms of a disease, gene therapy attacks a disease at its origin – the DNA of the patient. This type of gene therapy is known as somatic gene therapy, which targets the defect in the specific tissue that causes the disease of a patient. The other type of gene therapy is germ line therapy. Here, the sperm or eggs of a patient are treated in order to prevent a genetic disorder from affecting offspring. While these two types of gene therapy both accomplish the same goal of eradicating disease, they have drastically different implications. Somatic cell therapy only affects the treated patient. With germ line therapy, the results are much more extensive. The changes made in the sperm or eggs of the patient will not only affect their offspring, but can also affect future generations, thereby introducing a genetic change into the human population. One new gene therapy technique that is causing great concern in the scientific community is the Clustered Regularly Interspaced Short Palindromic Sequences, or CRISPR technique. This technique allows for highly specific gene manipulation, with DNA sequences able to be cut out or added with ease. The CRISPR technique uses an enzyme called Cas9 along with a strand of RNA that will guide it to the target sequence of DNA. Once at the desired DNA sequence, the Cas9 enzyme can edit the DNA or insert new sequences. The reason this technique causes concern is that it allows for the convenient and cheap modification of genes in cells, including sperm and eggs. While CRISPR has not been extensively tested in humans and human germ lines, the rapid growth and popularity of this technique raises concerns. One worry is that CRISPR could eventually be used to create heritable changes in the human genome by introducing genes and traits that did not exist before. Others worry that these genetic mutations could prove to be harmful, as it may disrupt the delicate balance of entire ecosystems through introducing genetically engineered organisms. This cutting edge technique will continue to be at the forefront of the ethics of gene therapy for years, and only time will tell where it will take the human population. Sources: [1] http://learn.genetics.utah.edu/content/genetherapy/gtintro/ [2] http://ghr.nlm.nih.gov/handbook/therapy/genetherapy [3] https://www.sciencebasedmedicine.org/crispr-and-the-ethics-of-gene-editing/ [4] https://www.ndsu.edu/pubweb/~mcclean/plsc431/students/bergeson.htm [5] http://www.nature.com/news/crispr-the-disruptor-1.17673 By Mae Grewal Writer  From: Charles River Laboratories From: Charles River Laboratories It’s now officially possible to grow a rat arm. Let that sink in for a moment: science and technology have progressed far enough that there is a way to induce cells to grow into the infinitesimally intricate, fully functioning, dismembered limb of a rodent. The project was undertaken by Harold Ott at Massachusetts General Hospital in Boston, and makes use of a process called decel-recel. In the decellularization step, the limb of a dead donor is treated with chemicals that strip away soft tissue, but leave the inert architectural protein matrix intact. This preserves the vastly complex structure of bones, blood vessels, muscles, and tendons that make up a functioning limb. Then, in recellularization, this scaffold is seeded with the appropriate cells from the eventual recipient of the biolimb. Because the new limb only contains cells from the recipient and not from a foreign source, the limb is much less likely to be rejected by the recipient.. Once Ott and his team grew the muscles of the new limb, they tested whether these muscles actually worked by electrically stimulating them. They found they could make the biolimb clench and unclench its paw, demonstrating successful muscle function. Furthermore, they tested the circulatory system by attaching biolimbs to anesthetized rats and observing whether or not the blood flowed adequately through the new vessels. This groundbreaking research puts medicine one step closer to providing amputees with another—possibly better—option in terms of regaining some of the functions of their lost limb. Current options include advanced prosthetics that allow for almost full range of motion but little to no sensory reception, and hand transplants, which run the risk of being rejected. If regrowing limbs from the amputees’ own cells proves viable, then having to choose between no touch or possible infection will become a thing of the past. Of course, there is still a ways to go in developing this technology for human use. The hand is one of the most complex parts of the body with its sensory and motor nerves and 1-cell-thick network of capillaries. While decel-recel regeneration has seen varying degrees of success with other organs such as the lungs, voice box, and kidneys, the increased complexity of the hand creates new challenges that need to be overcome. Most significantly, it remains to be seen whether a recipient’s nervous system will be able to grow into their new limb, and whether the new tiny capillaries will be able to maintain their structure to allow for blood flow without collapsing and creating clots. Ott himself says that it will be at least a decade before the first biolimbs are ready for human tests, but he, and many others, remain hopeful that technology will continue to progress to this eventual goal. In ten years, who knows? Forget rats, we could be growing human limbs in mass production for the benefit of trauma victims everywhere. By Sheela Ranganathan Writer Facebook, Instagram, Twitter, Yik Yak, Snapchat… Social media increasingly controls the lives of teenagers as these apps incessantly gain popularity and the convenient means to network progressively expand. Simultaneously, anxiety has become the greatest mental illness that affects college students in modern American society. I used to roll my eyes at my parents when they told me to limit my time on my phone and to share less posts to my ambiguous, large social media audience. However, researchers from the University of Scotland state that social media is directly linked to depression, anxiety, and poor sleep quality. In particular, those who logged on to these websites at later times in the night demonstrated greater likelihood of risks in mental health. In addition, receiving more or less likes on posts results in beneficial or adverse effects respectively. Extensive studies at the Freie Universität in Berlin demonstrated that receiving likes or tags on Facebook boosted the reward center in the brain. In addition to demonstrating the neurological effects of attention on the brain, this study also provided a basis for how posting on social media can become a compulsive addiction. So, the obsessive quest for compliments on pictures and posts is linked to patterns in the reward and pleasure pathways of the brain. Conversely, the lack of likes to one’s pictures results in feeling undesired or unnoticed. In addition, especially with respect to high school and college students, the concept of pictorial perfection leads to depression as people feel that their own lives are not as exciting or meaningful as the lives of others. Therefore, there is a direct correlation between the length of time spent on social media and the intensity of depression. To scroll or not to scroll? Despite these harms, social media definitely has many perks. However, if viewing the posts on your network makes you feel less confident or happy about yourself, it may be time to hit delete on these apps. Works Cited
[1] http://www.businessinsider.com/social-media-use-can-contribute-to-anxiety-and-depression-2015-6 [2] http://www.huffingtonpost.com/entry/social-media-college-mentalhealth_55ae6649e4b08f57d5d28845 [3] http://sundial.csun.edu/2015/03/to-like-or-not-to-like-how-social-media-effects-self-esteem/ [4] http://healthland.time.com/2013/08/31/this-is-your-brain-on-facebook/ [5] http://www.health24.com/Lifestyle/Teen/News/Social-media-may-increase-depression-in-teens-20150915 Thinking the Thought and Walking the Walk: How Technology is Helping Paraplegics to Walk Again12/2/2015 By Mae Grewal Writer Most people don’t really think about walking. When we want to get someplace, it just sort of happens—our brain send signals down our spinal cord into our peripheral nervous system to our motor nerves which cause our leg muscles to contract and suddenly we’re moving across the room to get to where we want to go without having to worry about the details, leaving our mind free to think about the essay that’s due in 12 hours or that cute guy at the RHO or the abysmal state of our political candidates for presidency. Unfortunately not everyone is this lucky. Fortunately, technology is evening the gap. Spinal cord injuries can render a person varying degrees of paralyzed, depending on at which vertebrae the injury occurred. Paraplegia is a result of a spinal cord injury that severs the nerves in the spinal cord rendering the bottom half of the body unable to move. Paraplegic patients have little hope of ever walking again without the assistance of expensive robotic limbs the route for the signal between their brain and legs is damaged. New research is looking to bypass that destroyed route entirely, however, by establishing a different way for the brain to relay walking signals to the leg muscles—and the crazy awesome thing? So far it’s working. A 26 year-old man was wheel chair bound for 5 years after his accident, but now he has become the first paraplegic to walk using impulses created from his own brain. He was outfitted with an EEG cap that monitored his brain waves and was able to distinguish between walking, sitting, and standing signals that the man actively thought. The cap then activates a electrical stimulator at his waist to create the muscle contractions to move the right leg forward, then the left, and so on, for an actual walking motion. He doesn’t have to actively think about each individual leg, but rather about the general motion of walking, and the waist monitor translates that into the more complicated individual muscle movements for him. But even this generalized thinking about walking took a lot of practice. While the brain does not lose the capacity to remember how to walk even if the body is paralyzed, his brain needed to be trained to actively voice that motion, which they practiced by hooking his EEG cap up to a computer avatar that he learned to control. Then, he needed to build up the muscles in his legs to support his own weight again, and finally, after 19 weeks, he was able to control his own movement and weight on a 3.5-meter course on the ground. While this alone is a breakthrough in the field of reestablishing function and control in handicapped persons, there is still a ways to go in perfecting this technology and making it accessible to all those who could benefit from it. The various machines that helped this man walk are quite unwieldy, and ideally should be minimized into implanted brain and muscle chips for maximum convenience. Furthermore, researchers are working on improving the precision of the computer system that recognized the will to move, as it currently still confuses walking intent with balancing intent, something our brains and nerves manage simultaneously. The potential is definitely there though to reintroduce the world of walking—and maybe even someday the world of walking without having to concentrate on it—to paraplegic patients. Works Cited
[1] http://www.theguardian.com/science/2015/sep/24/paraplegic-man-walks-with-own-legs-again |
AuthorsThe authors of these blog posts are staff writers of The Triple Helix at Georgetown University. Archives
November 2016
|



 RSS Feed
RSS Feed
